-
Ingredient SolutionsQuickly narrow your search. Browse products in our sortable catalog. View Catalog
-
ApplicationsQuickly narrow your search. Browse products in our sortable catalog. View Catalog
-
ResourcesRecently Posted
-
PLT People & Planet
-
About
Our international network, passionate team of experts and extensive industry knowledge is what sets us apart.
 Seth FlowermanCEO
Seth FlowermanCEO
SLENDACOR® Weight Management Complex
OVERVIEW
SLENDACOR® Weight Management Complex offers rapid weight loss, body shaping benefits and a thermogenic calorie burning benefit with a low, once per day dose.


.png)

12 lb. Average Weight Loss
People taking SLENDACOR lost an average of 12 pounds over an 8-week study.

6X More Effective Weight Reduction
SLENDACOR was shown to be 6 times more effective than diet & exercise alone in reducing weight.

4X Better BMI
Body Mass Index improvement was 4 times better with SLENDACOR than the placebo.

Up To 15% More Calories Burned At Rest*
SLENDACOR shows non-stimulant thermogenesis and increases resting metabolic rate.
Slendacor®: Clinically Proven Support for Healthy Weight Management
Discover Slendacor®, a clinically studied botanical blend designed to support healthy weight management. Backed by human clinical research, Slendacor helps promote fat loss, supports a healthy waistline, and fits seamlessly into modern weight management formulations.
Learn how Slendacor delivers results through science and why it’s trusted by brands worldwide.
Slendacor Clinical Study Video
Efficacy of a novel herbal formulation for weight loss demonstrated in a 16-week randomized, double blind, placebo-controlled clinical trial with healthy overweight adults. This video discusses the Slendacor clinical trial that was published in the journal Diabetes, Obesity and Metabolism in 2018.
Dixit K, et al. Efficacy of a novel herbal formulation for weight loss demonstrated in a 16-week randomized, double-blind, placebo-controlled clinical trial with healthy overweight adults. Diabetes Obes Metab. 2018;20(11):2633-2641. doi:10.1111/dom.13443 Video produced by researchsquare.com
FEATURES & BENEFITS
Three gold-standard clinical trials |
|
Statistically significant weight loss within 14 days |
|
Non-stimulant thermogenesis |
|
Increases Resting Metabolic Rate |
|
Helps burn up to 15% more calories at rest |
|
Significantly improved serum lipids |
|
Enhanced user mood |
|
Results seen in core: waist and hips |

CERTIFICATIONS
VEGAN
GLUTEN-FREE
NON-GMO
HALAL
KOSHER
An Innovative Botanical Complex
SLENDACOR is a novel, patented formulation of three standardized herbal extracts: turmeric (Curcuma longa), moringa (Moringa oleifera) and curry leaf (Murraya koenigii) that was developed through an extensive botanical screening effort that had the goal of identifying herbs that impact weight loss.
Curcumin
Curcumin is the principal curcuminoid of turmeric (Curcuma longa), a member of the ginger family.

Curry
Curry leaves come from the Murraya koenigii plant, which is native to the Indian subcontinent and Sri Lanka.
Drumstick Leaves
Common names include moringa, drumstick tree, horseradish tree, and ben oil tree or benzolive tree.
Rhodiola rosea
Rhodiola rosea is a plant native to remote Arctic climates in Asia, Europe and North America.

Exciting Phytochemistry
The root of the plant contains around 140 chemical compounds including phenols, rosavin, rosarin, salidroside and more.
Detail 1
Kale chips cold-pressed put a bird on it mumblecore kogi brooklyn farm-to-table blue bottle yuccie authentic kombucha migas. Literally tilde tacos paleo.

Detail 2
Kale chips cold-pressed put a bird on it mumblecore kogi brooklyn farm-to-table blue bottle yuccie authentic kombucha migas. Literally tilde tacos paleo.
RESEARCH
Research That Builds Trust With Customers
SLENDACOR has been studied in three human clinical trials to date – along with multiple preclinical studies. The design and results of this research offer a range of significant opportunities for your weight management product messaging. Study participants included both men and women. Subjects were of a broad age range and included a healthy population of overweight, not obese, individuals, adding confidence to results in the general population. Calorie-burning effects were seen on the first day of study. Significant weight loss was seen on Day 14, continuing throughout the duration of the study. The research shows statistically significant slimming—with a reduction in waist circumference and no reduction in lean body mass. Serum lipids and the Visceral Adiposity Index improved. In a 2021 study, calorie-burning effects were seen on the first day and the mood of study participants improved.
Clinical Science Demonstrates Weight Loss, Body Shaping, Calorie Burning and Enhanced Metabolism
Slendacor has been studied in three human clinical trials to date and multiple pre-clinical studies. A 2018 16-week double-blind, placebo-controlled study of Slendacor set new standards for communicating efficacy to consumers and regulatory agencies in the weight management category – because of its size, its length, its use of healthy, non-obese men and women, its design and execution. Consistent body weight reductions were observed throughout the 16-week trial – starting with statistically significant weight loss at two weeks. Participants averaged a total weight loss of nearly twelve pounds by the end of the study. The subjects taking Slendacor lost two inches in waist circumference and had no reduction in lean body mass.
New Studies Shows Thermogenic Benefits and Enhanced Metabolism
Two recent pre-clinical studies on energy expenditure showed that the mechanisms of action are related to the effect of Slendacor on white adipose tissue increasing UCP1 expression to resemble what occurs in more metabolically active brown adipose tissue. The 2021 human clinical trial (manuscript in development) was undertaken to further understand the thermogenic effects of Slendacor in human subjects.
A 2022 human clinical study (awaiting publication) focused on a single daily dose of Slendacor and its effects on resting metabolic rate. Participants using Slendacor showed up to a 15.2% increase in resting energy expenditure (REE) at Day 7. REE and calorie burning increased significantly from baseline on Day 1 of the study. The study also measured heart rate, blood pressure and mood. At no point during the study did heart rate or blood pressure increase in the Slendacor group, indicating there was no stimulant activity – unlike many other thermogenic ingredients.
The new data on lipolysis shows that Slendacor is not just breaking fat down but increasing the metabolism of that fat and changing the metabolic activity of those fat cells. It is burning fat rather than sending it to energy reserves. These new studies help understand Slendacor as an integral part of a holistic health management program, one that includes body weight management, but in the greater context of cardiometabolic health.
Mood and Psychological Well-being
The Profile of Mood States (POMS) is a psychological rating scale used to assess mood states. In clinical trials, participants taking Slendacor reported improvements in their POMS scores, indicating better mood and psychological well-being. This enhancement in mood can be attributed to the overall health benefits and metabolic improvements brought about by Slendacor.
CLINICAL HIGHLIGHTS
Clinical Study: Rapid weight loss and body shaping
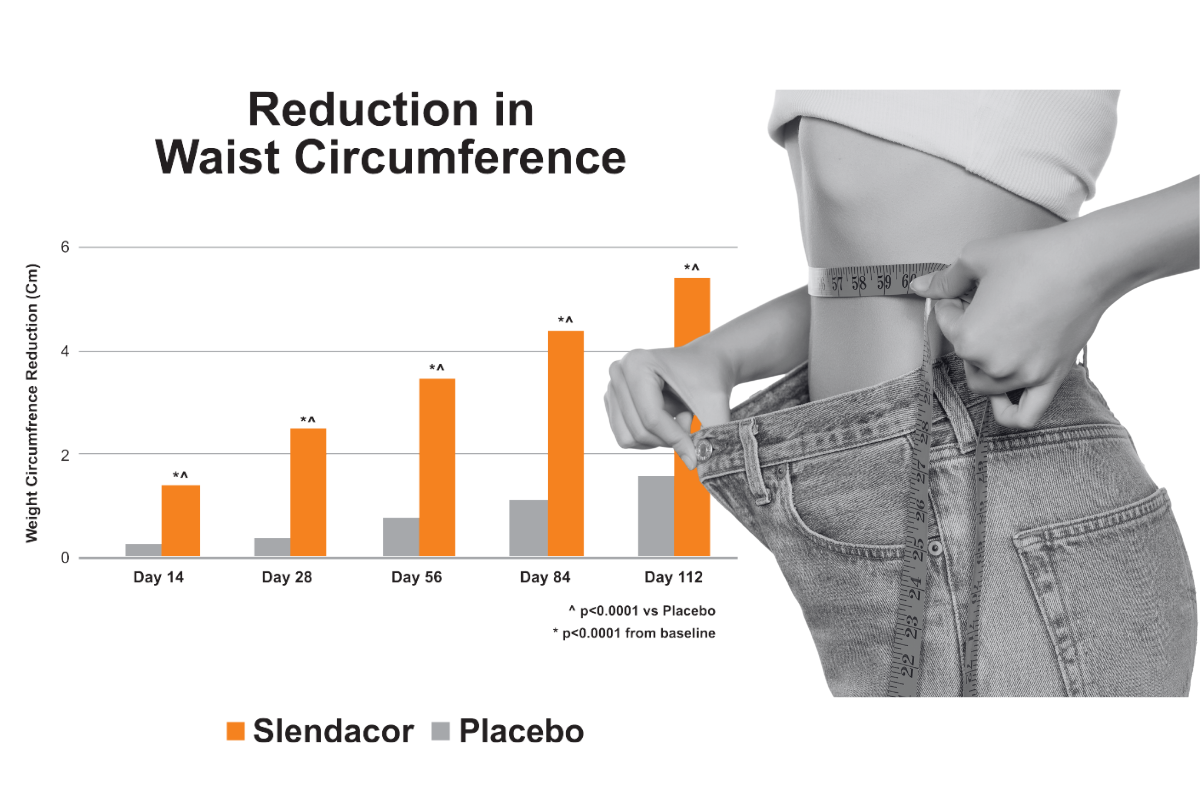
A 16-week double-blind, placebo-controlled study of Slendacor featured healthy, non-obese men and women. Consistent body weight reductions were observed throughout the 16-week trial – starting with statistically significant weight loss at two weeks. Participants averaged a total weight loss of nearly twelve pounds by the end of the study. The subjects taking Slendacor lost two inches in waist circumference and had no reduction in lean body mass.
Dixit K, et al. Diabetes Obes Metab. 2018 Nov;20(11):2633-2641.)

Clinical Study: Weight loss, thermogenic benefits and enhanced metabolism
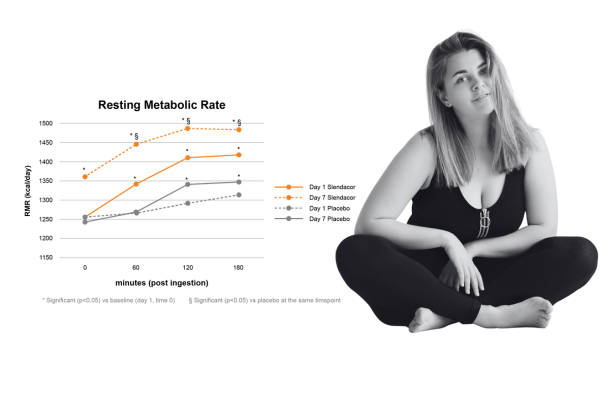
A 2022 human clinical study (awaiting publication) focused on a single daily dose of SLENDACOR and its effects on resting metabolic rate. Participants using Slendacor showed up to a 15.2% increase in resting energy expenditure (REE) at Day 7. REE and calorie burning increased significantly from baseline on Day 1 of the study. The study also measured heart rate, blood pressure and mood. At no point during the study did heart rate or blood pressure increase in the SENDACOR group, indicating there was no stimulant activity – unlike many other thermogenic ingredients. Salter D, Murphy JM. A blend of Curcuma longa, Moringa oleifera, and Murraya koenigii extracts elevates resting metabolic rate without changes to heart rate or blood pressure.
Curr Develop Nutr 2023 [in press]Dixit K, et al. Diabetes Obes Metab. 2018 Nov;20(11):2633-2641.)

MARKET OPPORTUNITIES
Weight Management
SLENDACOR has helped power some of the world’s most successful weight management brands used in product forms from powders and shakes to chews and shots.
Active/Sports Nutrition
SLENDACOR is perfect for adding a weight management benefit to active and sports nutrition products.
Bone, Joint & Muscle Health
Being overweight/obese is a contributing factor to the development of joint pain. SLENDACOR can help your formulation.
Healthy Aging
Studies have shown that SLENDACOR can support cardiovascular and metabolic health. It’s a healthy approach to weight management.
APPLICATIONS

BEVERAGES

CHEWS

GUMMIES
EFFERVESCENTS

POWDERS

RTDS/SHOTS

SOFT GELS

STICK PACKS

TABLETS
ORIGIN STORY
An Intensive Search of Traditional Botanical Medicine
The development of SLENDACOR paired traditional botanical medicine with modern scientific evaluation – through a directed program of screening. It examined thousands of plant extracts for their ability to stop fat cells from maturing - a process known as ‘adipogenesis.’

Three Botanicals
Out of these thousand, three of these botanicals showed the best anti-adipogenesis activity. Curcuma longa – also known as curcumin or turmeric; Moringa oleifera – or Drumstick leaf - and Murraya koenigii also known as curry leaf.

Combination for Lipolysis
The next step examined different ratios of the three botanicals to see whether together, they could offer even better performance. One combination offered the strongest potential for preventing fat cell maturation and was also shown to increase fat breakdown (lipolysis).

Three Human Clinical Trials
The next phase of the screening process evaluated whether this combination would enhance weight loss when taken as a supplement. Testing involved animal studies and three human clinical trials.

Positive Results: SLENDACOR
The results were overwhelmingly positive. The clinical studies demonstrated that our newly discovered ratio offered rapid, consistent weight loss – and the ingredient we know today as SLENDACOR was born.

INSIGHTS
Water dispersible takes you beyond the pill
There’s a reason we talk about ‘pill fatigue’ today. People can only take so many of them. Increasingly, consumers are looking to get the nutrition and support they want from a diverse range of product forms that are fun and convenient. The water-dispersible grade of SLENDACOR delivers the power of the traditional ingredient in a neutral-tasting form which means you can use it in functional foods as well as a broad range of supplement delivery systems.
SLENDACOR recognized twice as Weight Management Ingredient of the Year
When SLENDACOR Weight Management Complex was first introduced in 2017, it was awarded the Weight Management Ingredient of the Year by Nutraingredients based on the powerful science represented in its second clinical trial showing weight loss starting at 14 days, reduction in BMI and reduction in waist and hip circumference.
Five years later, SLENDACOR won the same award based on the cutting-edge clinical trial that examined resting metabolic rate, non-stimulant thermogenesis and calorie burning increased by up to 15% starting almost immediately upon supplementation.
A major research effort went into creating a new form of SLENDACOR that delivers the power of the traditional ingredient in a neutral-tasting, water-dispersible form. Allowing it to be used in functional foods as well as a broad range of supplement delivery systems.


“Our new data on lipolysis shows that SLENDACOR is not just breaking fat down but increasing the metabolism of that fat and changing the metabolic activity of those fat cells.”
Dr. Jeremy Appleton, ND, Director of Medical and Scientific Affairs for PLT
RESOURCES


Behind every ingredient solution is an even better story, download the product sheet to learn more.
Explore the latest in cognitive support for well-being with PLT's ingredient platform. In this webinar, you'll discover the science behind cognitive ingredients and how to incorporate them into formulations.











Neutral Tasting, Water-Dispersible Form of Industry Leading Weight Management Ingredient Will Allow Product Formulators to ‘Go Beyond the Pill’.







PLT Health Solutions reveals an expansion of its intellectual property with a new patent for its Slendacor weight management complex. According to the company, the patent is based on recent clinical research showing how Slendacor’s thermogenic properties promote weight loss and body shaping.
Help Your Customers Discover a Natural Alternative to Weight Loss Drugs
Are you ready for SLENDACOR to power your brands?
SLENDACOR Weight Management Complex offers a broad range of weight management messaging opportunities – backed by clinically studied performance. Ask us how it can help you.
- Expertise
- Ingredient Solutions
- All
- Animal Health & Wellness
- Beauty from Within
- Joint & Bone
- Cardiovascular
- Cognitive Performance
- Energy
- Functional Foods & Beverages
- Healthy Aging & Longevity
- Hydration+
- Immune & Respiratory Health
- Men’s Health
- Muscle Health
- Pain & Mobility
- Plant-Based Nutrition
- Sexual Health
- Sleep
- Sports & Active Nutrition
- Stress & Mood
- Weight Management
- Women’s Health
- Applications
- Resources
- PLT People & Planet
- About
These products are not intended to diagnose, treat, cure or prevent disease. This website is for informational purposes only.














